Learn Drug regulatory authorities in pharmacy with list. Explore definition, list and key roles of these authorities.
Each country has a regulatory agency that is accountable for imposing rules and regulations. It passes guidelines for drug development, registration, licensing, manufacturing, marketing and labelling of pharmaceutical products.
Drug regulatory authorities
Drug regulatory authorities define as organizations that ensure medicines are safe and effective for public use, overseeing their approval, quality and safety. Regulatory authorities are being established in different countries all over the world.
Pan American Health Organization (PAHO), International Conference on Harmonization (ICH), World Health Organization (WHO), World Trade Organization (WTO) and World Intellectual Property Organization (WIPO) are various international regulatory authorities and organizations that are important for all facets of pharmaceutical regulations associated to drug product registration, manufacturing, supply, price control, marketing, research and development.
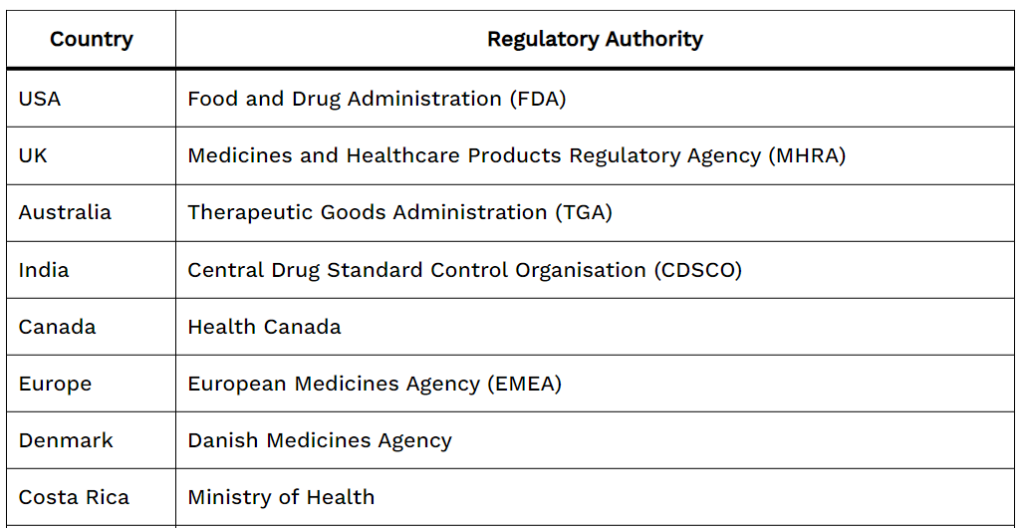
List of Drug regulatory authorities
| Country | Drug regulatory Authority |
| USA | Food and Drug Administration (FDA) |
| UK | Medicines and Healthcare Products Regulatory Agency (MHRA) |
| Australia | Therapeutic Goods Administration (TGA) |
| India | Central Drug Standard Control Organisation (CDSCO) |
| Canada | Health Canada |
| Europe | European Medicines Agency (EMEA) |
| Denmark | Danish Medicines Agency |
| Costa Rica | Ministry of Health |
| New Zealand | Medsafe – Medicines and Medical Devices Safety Authority |
| Sweden | Medical Products Agency (MPA) |
| Netherlands | Medical Evaluation Board |
| Ireland | Irish Medicines Board |
| Italy | Italian Pharmaceutical Agency |
| Nigeria | National Agency for Food and Drug Administration and Control (NAFDAC) |
| Ukraine | Ministry of Health |
| Singapore | Centre for Pharmaceutical Administration, Health Sciences Authority |
| Hong Kong | Department of Health – Pharmaceutical Services |
| Paraguay | Ministry of Health |
| Thailand | Ministry of Public Health |
| China | State Food and Drug Administration |
| Germany | Federal Institute for Drugs and Medical Devices |
| Malaysia | National Pharmaceutical Control Bureau, Ministry of Health |
| Pakistan | Drugs Control Organisation, Ministry of Health |
| South Africa | Medicines Control Council |
| Sri Lanka | SPC, Ministry of Health |
| Switzerland | Swissmedic, Swiss Agency for Therapeutic Products |
| Uganda | Uganda National Council for Science and Technology (UNCST) |
| Brazil | Agencia Nacional de Vigilancia Sanitaria (ANVISA) |
| Japan | Ministry of Health, Labour and Welfare (MHLW) |
Role
Key roles of regulatory authorities are:
- Safety assurance is a key role.
- Monitoring quality during manufacturing.
- Overseeing clinical trials for safety.
- Verifying accurate drug labels.
- Investigating adverse drug reactions.
- Controlling drug pricing if needed.
- Protecting intellectual property.
- Educating the public on safe drug use.
- Enforcing rules and recalls when necessary.
You may also like to read Regulatory Affairs in Pharma.
Short Answer Questions (SAQ)
- Question: What is the primary responsibility of drug regulatory authorities?
Answer: Drug regulatory authorities ensure that medicines are safe and effective for public use. - Question: Which country’s regulatory authority is known as the FDA, and what does it stand for?
Answer: The USA has the FDA (Food and Drug Administration). - Question: What is the role of International Conference on Harmonization (ICH)? Answer: The ICH develops and harmonizes guidelines for drug registration globally.
- Question: Why do regulatory authorities oversee clinical trials?
Answer: Regulatory authorities oversee clinical trials to ensure safety and ethical conduct. - Question: What role do regulatory authorities play in monitoring drug labels?
Answer: They verify that drug labels provide accurate information for healthcare professionals and patients. - Question: In which country is the Central Drug Standard Control Organization (CDSCO) located?
Answer: CDSCO is located in India. - Question: What is the key role of regulatory authorities in protecting intellectual property?
Answer: They safeguard pharmaceutical patents and trademarks. - Question: What is the primary goal of drug pricing control by some regulatory authorities? Answer: To ensure the affordability of medicines.
- Question: What is the significance of regulatory authorities in enforcing rules and recalls? Answer: They enforce compliance through inspections and recalls when necessary to protect public health.
